Traditional 2D cultures have been used widely over the past decades to study cell biology, molecular biology and conduct translation research such as drug discovery. Cells in 2D culture, however, are forced to adopt a planar morphology and maintain cellular interactions only in lateral directions, altering gene transcription, protein translation, and functional phenotypes. As a result, there is a shift towards using 3D in vitro models in the last several years as cell morphology and physiology more closely represent cells in vivo.
There are 2 main types of 3D culture systems known as scaffold-based and scaffold-free. In Table 1 below, the advantages and disadvantages of the different 3D cell culture techniques are listed to help researchers determine the most appropriate 3D culture method for their research.
Table 1: Advantages and Disadvantages of Different 3D Cell Culture Techniques.
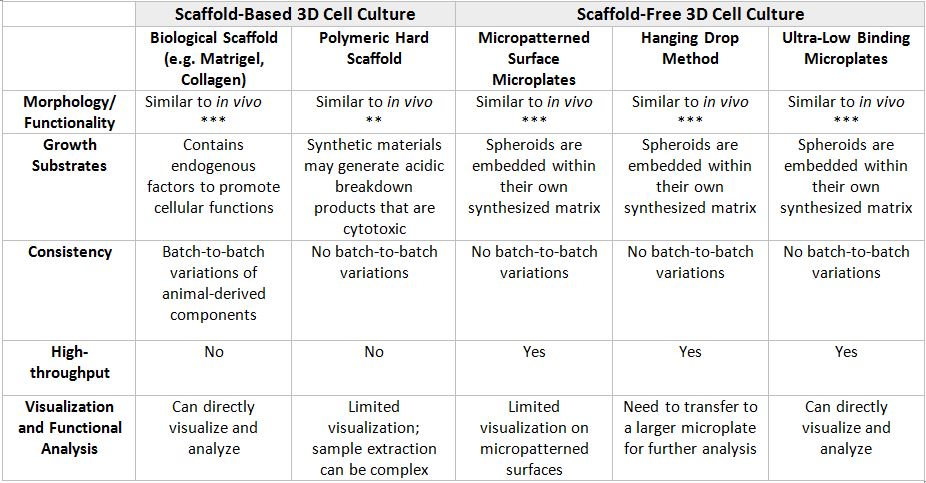
Due to their novelties and complexities, 3D cell culture technologies may be daunting to some researchers. Additionally, optimizing the many variables in 3D cell cultures can be costly and time-consuming. Nevertheless, the data and biological insights gained from the correct 3D cell culture models is invaluable. For example, 3D liver cell spheroid cultures are valuable for examining drug-induced liver toxicity, function, and disease. Human primary hepatocyte spheroids can self-assemble in low adhesion conditions and be phenotypically stable. Additionally, 3D spheroids retain hepatocyte-specific functions for at least 5 weeks, enabling chronic toxicity assessment of drug molecules. Notably, the chronic toxicity of fialuridine was detected after repeated dosing using a spheroid model, while 2D primary hepatocyte culture was unable to provide this integral information. Moreover, improved cell-specific function and gene expression have been observed in many different cell types including chondrocytes, osteoblasts, hepatocytes, myocytes, neurons, mammary, lung, and kidney epithelial cells under 3D cell culture conditions.
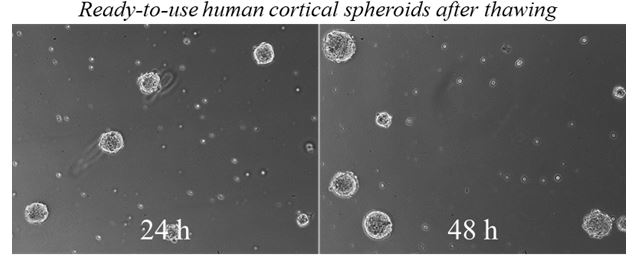
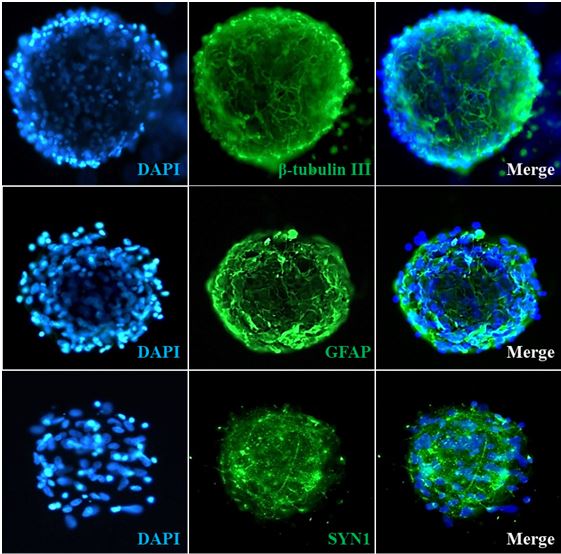
Given the primary cell resources and expertise we have, ScienCell has developed the highly innovative ready-to-use 3D primary spheroids to study cell biology and physiology under more relevant conditions. During embryonic development, cells undergo biological self-assembly to form 3D complex tissues through the cell-cell and cell-matrix interactions. Similarly, ScienCell’s ready-to-use spheroids maintain the extensive cell-cell and cell-matrix interactions. Spheroids are surrounded by their own secreted extracellular matrix that regulates cell proliferation, differentiation, and migration. Importantly, the sizes of the ready-to-use 3D spheroids are maintained around a few hundred micrometers to prevent necrotic damage to the core of the spheroid. Last but not least, each cryopreserved spheroid vial contains more than 10,000 spheroids, which lead to the formation of hundreds of 3D spheroids per well, enhancing the statistical power and the accuracy of your experimental data.
Experimental Timeline Using the ScienCell’s Ready-To-Use Spheroids
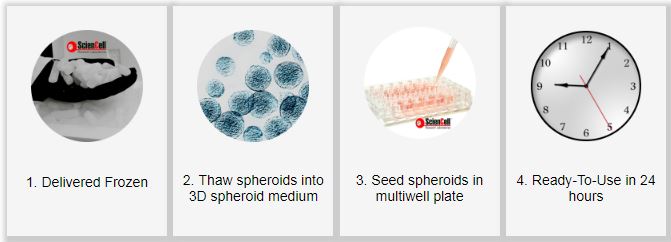
ScienCell offers human and rat ready-to-use 3D spheroids which are ready for your experiment within 24-72 hours after thawing. Currently, we offer the following models: blood-brain-barrier model, cerebral cortex model, vascularized bone model, and cartilage model.

Can 3D sphere blood brain barrier (BBB) model be used to test BBB drug penetration? if yes, could you explain how to use the sphere cultures to test BBB penetration by a drug?
Thank you
Kris
Hi Kris,
Yes, ScienCell’s 3D BBB spheroids can be used to study BBB drug penetration. The drug concentration present within the spheroids (which need to be lyzed) can be measured and normalized it to the drug concentration in the medium supernatant. Thanks!